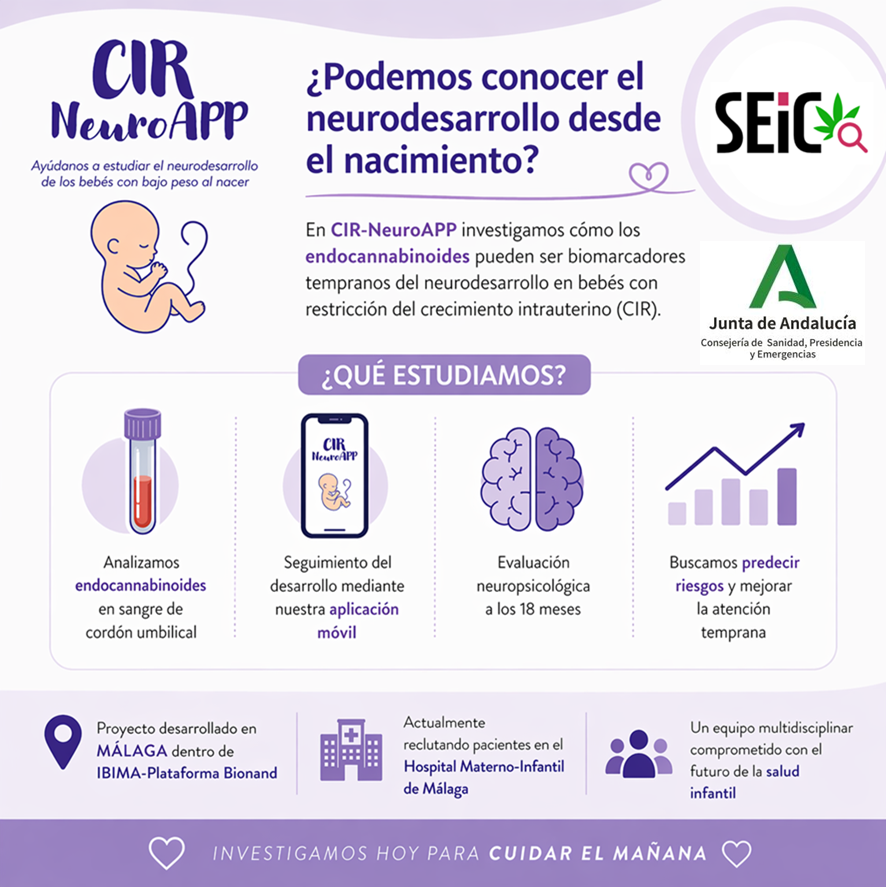
The CIR-NeuroAPP project is a research initiative focused on improving the early detection of neurodevelopmental alterations in children born with intrauterine growth restriction (IUGR), a condition affecting approximately 10% of pregnancies and associated with an increased risk of short- and long-term neurological complications.
One of the main clinical challenges in this field is identifying which newborns are at higher risk of developing cognitive, motor, or behavioral difficulties. To address this challenge, CIR-NeuroAPP proposes an innovative approach based on the integration of biomarkers with a follow-up mobile application.
CIR-NeuroAPP (PI-0070-2025) has been funded by the Regional Ministry of Health, Presidency and Emergencies of the Government of Andalusia and is led by SEIC-affiliated researchers Dr. Marialuisa Francesca de Ceglia and Dr. Patricia Rivera González (IBIMA and University of Málaga). The project involves a multidisciplinary team of experts in neuroscience, obstetrics, neonatology, and clinical psychology. It is currently in the active participant recruitment phase at the Mother-and-Child Hospital of Málaga.
The role of the endocannabinoid system
Among the biomarkers under study, the project places special emphasis on endocannabinoids, key lipid mediators involved in processes such as neurogenesis, synaptic plasticity, inflammatory responses, and stress adaptation. In addition, the endocannabinoid system plays a fundamental role during pregnancy by regulating processes such as implantation, placental development, and nutrient transport to the fetus.
The project’s central hypothesis is that alterations in endocannabinoid levels in umbilical cord blood may reflect early dysfunctions in brain development, thereby serving as predictive biomarkers of neurodevelopmental outcomes in children with IUGR.
An integrated approach: biomarkers + mobile technology
The project combines:
- Biomarker analysis at birth, including proteins associated with neuronal damage (S100B, NSE, tau protein), neurotrophic factors (NGF, BDNF), and endocannabinoids.
- Longitudinal follow-up through a mobile application (CIR-NeuroAPP) that enables families to easily record early childhood developmental milestones.
- In-person neuropsychological assessment at 18 months of age, to validate the predictive capacity of the biomarkers.
This integrated approach will enable the development of an early predictive model, facilitating the identification of children at risk of adverse neurodevelopment and promoting timely, personalized early interventions.